Cell and gene therapies (CGTs) represent one of the most profound shifts in modern medicine, offering the potential for durable – and in some cases curative – outcomes for patients with severe, previously intractable diseases. Early CGT launches demonstrated the power of this promise. Several first-in-class therapies achieved strong initial uptake, driven by transformative efficacy, compelling clinical value propositions, and clearly identifiable patient populations.
However, as the CGT market has entered the next phase of growth, commercial performance has become more uneven. While some therapies continue to show robust demand, others – despite strong science – have struggled to gain traction due to durability concerns, payer resistance, logistical and operational bottlenecks, restricted eligible populations tied to safety or label limitations, and/or lower than anticipated demand. In some cases, therapies with regulatory approval have even been withdrawn from the market due to insufficient commercial uptake.
These challenges are not primarily failures of innovation, but often failures of execution within an extraordinarily complex commercialization environment. In addition to complexities faced in traditional pharmaceutical launches, CGTs require orchestration across a highly fragmented and interdependent journey that spans:
- Early symptom recognition and diagnosis
- Molecular and biomarker testing
- Treatment option evaluation with specialists and genetic counselors
- Referral to an Authorized Treatment Center (ATC)
- Manufacturing and cold-chain logistics
- Benefits verification, prior authorization, and payer negotiation
- Pre-conditioning, infusion, monitoring, and long-term follow-up
Each step in this pathway introduces potential points of friction where patients, momentum, and value can be lost. These “leakage points” are magnified by the emotional weight of life-altering diagnoses, the novelty of the technology, and the economic implications of one-time, high-cost treatments.
As a result, CGT commercialization cannot rely on conventional pharmaceutical launch playbooks. Instead, success depends on early, coordinated investment across medical, operational, access, and commercial domains, combined with a deep understanding of patient, provider, and payer beliefs, behavior and decision processes within this complex ecosystem.
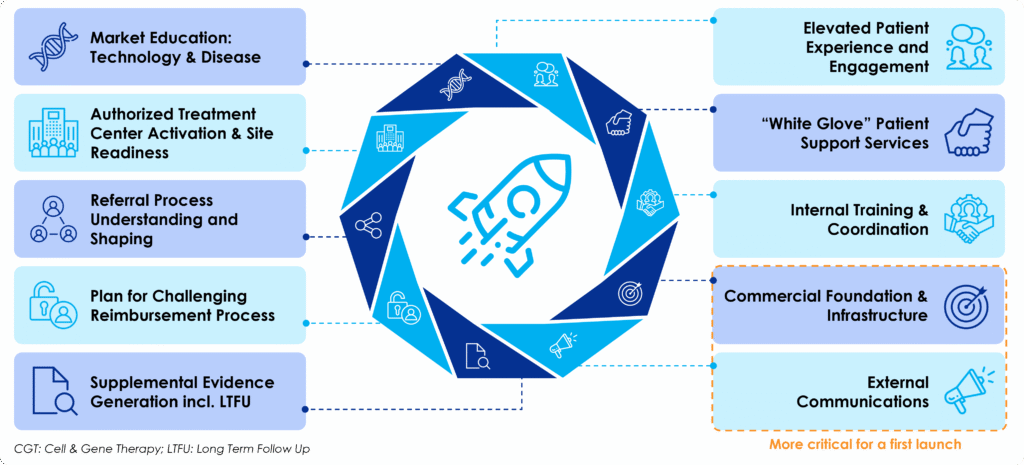
Based on observed launch experience and market dynamics, ten commercialization success factors consistently distinguish CGT therapies that achieve sustainable adoption from those that struggle.
Overview of 10 Critical Success Factors for Cell / Gene Therapy Launch & Commercialization
Now, let’s explore each of these success factors individually. In this paper, we provide a brief introduction and overview for each. In upcoming publications, we’ll tackle each one in considerably more detail, providing additional insights to biopharma companies that are operating – or plan to operate – in this space.
-
Market Education on Both Technology and Disease
CGT success requires sustained education on two distinct but equally important dimensions: the disease and the technology.
While disease education remains essential, particularly in rare or underdiagnosed conditions, technology education plays a uniquely critical role in CGTs. Stakeholder hesitancy often stems from limited familiarity with mechanisms of action, concerns about long-term safety, and uncertainty regarding durability of effect. Without proactive education, novel therapies risk being grouped together with broader market failures or misconceptions about the category as a whole.
Successful launches invest in technology education early, often three or more years before anticipated approval. These efforts build scientific credibility, establish differentiation, and help shape perceptions before payer and provider narratives become entrenched. Importantly, education on innovation alone is insufficient to drive demand; it must be paired with traditional disease awareness, patient-finding initiatives, and practical guidance on identifying appropriate candidates for therapy.
-
Authorized Treatment Center Activation and Site Readiness
Authorized Treatment Centers (ATCs) form the operational backbone of CGT delivery. However, companies frequently underestimate the effort needed to ensure their readiness.
Transitioning a site from a sponsor-supported clinical trial environment to a commercial ATC requires far more than certification. It involves developing new SOPs, implementing billing and reimbursement workflows, coordinating with payers, training multidisciplinary care teams, and integrating complex logistics and manufacturing schedules into routine operations.
ATC on-boarding is a marathon, not a sprint. Site selection, engagement, contracting, and SOP development typically requires 10–12 months of lead time (or more) and demands substantial financial and personnel investment. Successful activation depends on close coordination across medical, commercial, and operational functions, with clear prioritization of sites based on patient access, institutional readiness, and referral potential. Overall, this is a coordinated effort where field teams prepare the care team operationally, paving the way for commercial teams to facilitate the buying process.
It’s important to remember that when ATCs are not correctly prepared at launch, even strong clinical demand likely won’t translate into real-world uptake.
-
Referral Process Understanding and Shaping
The CGT referral pathway is rarely direct. Patients often move from primary care to community specialists before eventually reaching treatment centers authorized to administer therapy. Each handoff introduces the risk of delay, fatigue, or abandonment.
Referring physicians may hesitate due to uncertainty about patient eligibility, concern over financial implications, fear of losing long-term care relationships, or lack of clarity on how to initiate referrals. Without proactive guidance, these uncertainties can stall patient flow even when the clinical need is obvious.
Manufacturers that map real-world referral pathways and actively address these friction points, through education, peer-to-peer engagement, concierge referral hubs, and return-to-community care toolkits, are better positioned to preserve patient momentum. Peer mentoring programs linking community physicians with ATC specialists can be particularly effective in building confidence and normalizing referral behavior.
-
Planning for a Challenging Reimbursement Process
CGT reimbursement introduces economic and operational complexity well beyond standard specialty drugs. Payers are increasingly cautious about high upfront costs, particularly in the absence of long-term durability data. At the same time, treatment centers face substantial administrative burden in documenting eligibility, negotiating case rates, managing prior authorizations, and navigating appeals.
Therefore, commercial success depends on solving not only for payer coverage, but for institutional viability. Manufacturers that appropriately support ATCs in negotiating rates, managing workflows, engaging the “right” individuals at payers, and minimizing financial risk are more likely to achieve consistent access.
In addition, traditional pricing and contracting approaches often fail when applied to one-time therapies. That reality drives the need for creative and tailored strategies that address both payer skepticism and provider economics.
-
Supplemental Evidence Generation, Including Long-Term Follow-Up
Data collection doesn’t stop at approval. Early investment in post-launch evidence generation is essential to support downstream pricing discussions, payer dossiers, and economic value models. Key evidence needs include demonstration of unmet need, treatment burden, long-term safety, and durability of response. Cost-effectiveness and budget impact models must be available at launch and refined as real-world data accumulate.
It’s also wise to investigate and potentially engage in Value-Based Contracts (VBCs). While VBCs sound sensible for high-cost therapies, they are operationally complicated to implement. Long-term data collection partnerships between payers and healthcare delivery networks must be established early on.
In many cases, long-term follow-up registries and real-world evidence partnerships become foundational components of commercialization strategy. These efforts not only support access negotiations but also reinforce confidence among providers and patients navigating uncertainty around long-term outcomes.
-
Elevated Patient Experience and Engagement
The CGT patient journey is emotionally and logistically demanding. Delays, inconsistent terminology, misinformation, and fragmented communication can compound fear and confusion for patients and caregivers.
Effective engagement can help counteract some of the emotional and logistical drain. It requires mapping the full ecosystem, not only the patient, but caregivers, patient advocacy groups (PAGs), peer networks, providers, and payers. Manufacturers can play a constructive role by providing clarity around risks and benefits, countering misinformation, and helping patients navigate the system from diagnosis to infusion to long-term follow-ups.
Streamlining touchpoints across sites of care and harmonizing language and expectations improves continuity and reduces attrition during critical decision-making moments.
-
“White Glove” Patient Support Services
Standard hub services are insufficient for CGTs. These therapies demand highly customized, high-touch support models designed around specific disease states and institutional workflows.
Effective programs enable community to ATC referrals and logistics, ATC scheduling and procedures, financial complexity, and geographic constraints. Services often extend beyond reimbursement support to include travel coordination, manufacturing and shipment tracking, and proactive communication during the waiting period between enrollment and infusion.
This “white glove” approach not only improves operational efficiency but also addresses one of the most stressful phases of the CGT journey: the period of uncertainty while therapy is being manufactured and scheduled.
-
Internal Training and Cross-Functional Coordination
CGTs raise the bar for field team expertise. All external facing team members must demonstrate deep scientific and operational understanding to establish credibility and trust with a range of key stakeholders.
This often necessitates extended training timelines and specialized roles such as patient educators or access experts. Internally, cross-functional coordination is critical. Medical, commercial, and access teams must operate with shared strategy, consistent messaging, and clearly defined responsibilities.
Failures in handoff or misalignment between functions can disrupt relationships with key stakeholders and undermine trust at precisely the moment when coordination is needed the most.
-
Commercial Foundation and Infrastructure
Commercial planning for CGTs must begin two to three years pre-launch, building upon early medical and scientific groundwork.
Early market research informs understanding of stakeholder needs and shapes downstream investment. Infrastructure must support real-time performance tracking and rapid course correction, as small patient populations and complex logistics make launches particularly fragile.
Go-to-market models must also be adapted for one-time therapies. Field team sizing, incentive structures, and forecasting approaches must account for declining eligible populations over time and the absence of repeat prescribing behavior.
-
External Communications
CGT launches rarely follow conventional trajectories. Early uptake is often constrained by site readiness, payer negotiations, and operational throughput rather than lack of clinical interest.
Pre-launch communications should focus on technology credibility and infrastructure readiness to establish confidence. Post-launch communications should then shift toward leading indicators, such as metrics regarding site activations and intake initiations, and coverage metrics as the supporting key performance indicators (KPIs).
By establishing leading indicators and aligning forecasting assumptions early, manufacturers can manage expectations and maintain confidence among investors and stakeholders during the inherently slower ramp-up phase of CGT commercialization.
Thinking Ahead
Cell and gene therapies are redefining therapeutic possibilities, but their commercial success is not guaranteed by scientific excellence alone. The CGT journey introduces multiple interdependent points of friction across diagnosis, referral, reimbursement, and delivery.
Manufacturers that succeed are those that recognize commercialization as a systems challenge rather than just a promotional one. By investing early, coordinating across functions, and designing strategies grounded in real-world patient and institutional behavior, companies can translate scientific breakthroughs into sustainable patient access. Excellence in CGT commercialization is achieved not through a single innovation, but through disciplined execution across these ten critical success factors.
If you would like to connect with Blue Matter to discuss your organization’s goals and needs regarding CGT launch and commercialization, please contact us via our website. In addition, check our Insights page regularly or follow us on LinkedIn to stay up-to-date on our latest publications. Over the next few months, we’ll be diving deeper into each of the ten success factors described above.