New Ideas.
Better Results.
Blue Matter is a consulting firm serving the life sciences industry. We strive to bring a new approach to consulting with original ideas that deliver a meaningful impact.
Our Purpose
We are proud to be part of an industry that is pushing the boundaries of science and technology to improve people’s lives. Our role is to help our clients identify unique insights, generate new ideas and drive meaningful results to get the most out of their products, portfolios and teams.
We began our careers in established consulting firms where we developed valuable experience and industry knowledge. During this time, we came to see that rigid processes often interfered with original thinking and led to bloated deliverables packed with mediocre answers.
A group of us like-minded, passionate consultants believed there was a better approach and founded Blue Matter to provide a platform to foster innovation and creativity in consulting.
Our Difference
Industry and scientific knowledge, analysis and insights. Left brain thinking. This is the foundation of our discipline. Creativity and imagination. Constantly challenging conventional thinking and seeking fresh ideas. Right brain thinking. We’re passionate about this; it’s the reason our clients come back.
The key to winning is effective differentiation. And effective differentiation comes from originality–thinking that is grounded in reason and infused with imagination.
"The terrific work you did resulted in a substantial change to our commercial strategy."
- Chairman of the Board, Small Biotech
"Thank you very much for the great work and partnership. You are one of the rare breed of consultants whom I trust. You will always be on my speed dial if we need help with anything."
- Chief Commercial Officer, Small Biotech
"I appreciate all of the great work you have done for our team this past year and look forward to continued partnership in the New Year."
- Chief Commercial Officer, Mid-sized Diagnostic Company
"Your preparation and support has been instrumental in achieving our desired outcome."
- Franchise Commercial Lead, Global Biotech
"Thank you all so much for the extremely successful project...I couldn't be happier with the way that it went."
- Sr. Product Manager, Diagnostic Company
"This project was excellent, from the content development to facilitation. Many, many thanks for the great work!"
- Director of Marketing, Diagnostic Company
"The process was great, I loved that I didn't have to manage you that much. Everything was highly organized and done on time. Quality and originality of thinking was also good."
- Global Commercial Lead, Large Biotech
"Great project. I was learning as we went along; good expertise — brought in examples from other projects."
- Global Commercial Lead, Large Biotech
Our Clients
Our clients include: top global bio-pharmaceutical companies with large, diverse product portfolios to small emerging players with one asset in development, leading diagnostic companies, and investment houses. We work with CEOs, Chief Commercial Officers, Franchise Heads, Product Directors, Functional Area Leaders. 90%+ of our business is from repeat, satisfied customers.
Our Capabilities
Specialized Expertise, Integrated Strategy
With in-depth expertise in a range of areas, we deliver specialized support as well as integrated cross-functional strategies.
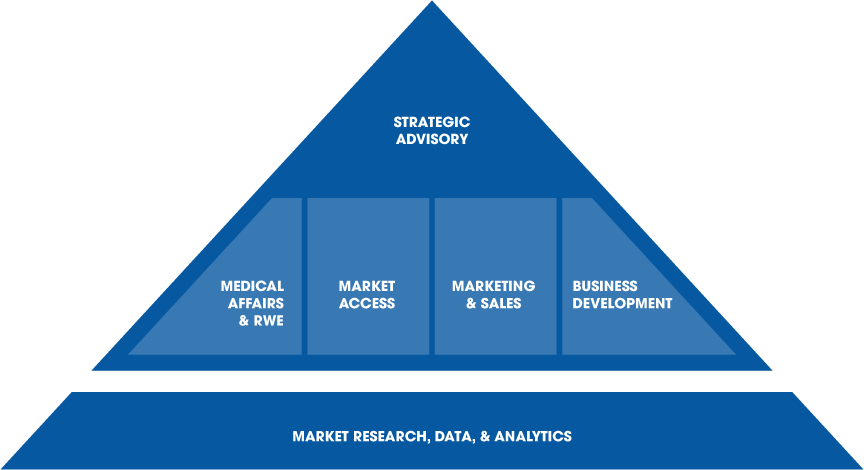
Comprehensive Services
We support clients across the product and company lifecycle, maximizing value at the product, portfolio, and organization levels.
Therapeutic Expertise
Broad therapeutic expertise with an in-depth focus on complex and specialty markets
Want to Join Our Team?
Blue Matter offers enriching project experiences with a diverse range of strategic, client-facing projects in the exciting and innovative life sciences industry.
Insights
The “Best Practice Organization,” Part 4: Ways of Working
Dispatch from MAPS 2024
The “Best Practice Organization,” Part 3: The Role of Capabilities in High-Performing Companies
Oncology Market Outlook for 2024
The “Best Practice Organization,” Part 2: How Vision, Mission, and Values Guide Biopharma Companies’ Actions and Underpin Their Success
Blue Matter Further Expands Analytics Team, Opens New Office in Gurugram, India
Hematologic Heavyweights: CAR-Ts & T-Cell Engager Bispecifics Are Primed for a Showdown
Companies
The Blue Matter family also includes other leading companies that provide specialized support to the biopharmaceutical industry.
Blue Matter companies often work together to help clients achieve their goals.












































